Theranostic
Theranostic Nucleosides as Personalized Medicines
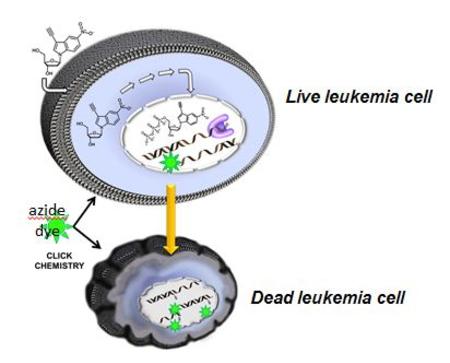 Personalized medicine emphasizes the customization of pateint care by tailoring treatments and therapeutic practices to an individual. Conventional approaches typically involve the molecular profiling of a patient’s cancer through genetic, proteomic, and/or metabolomic analyses. Unfortunately, these efforts have met with limited success.
Personalized medicine emphasizes the customization of pateint care by tailoring treatments and therapeutic practices to an individual. Conventional approaches typically involve the molecular profiling of a patient’s cancer through genetic, proteomic, and/or metabolomic analyses. Unfortunately, these efforts have met with limited success.
To combat this problem, Red5 Pharmaceuticals is developing more effective personalized treatments against cancer. Our research focuses on the development and applications of “theranostic” agents as anti-cancer agents. “Theranostic” agents are chemical entities that combine a specific therapeutic activity with unique diagnostic properties. Combining these properties provides physicians the ability to accurately measure the therapeutic activity of the drug in real time. This technology represents a new paradigm in personalized medicine as the proper therapeutic dose can be directly based on the behavior of the drug within an patient.
Background
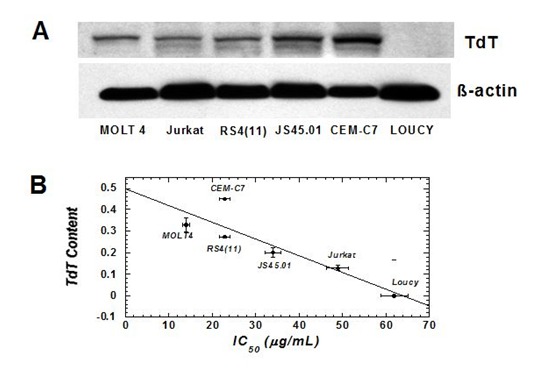
Red 5 has developed a small molecule that represents the first-in-class theranostic agent to treat acute lymphoblastic leukemia (ALL). ALL is the most common type of childhood cancer, presenting with approximately 5,000 new cases each year in the United States. Although the cause of ALL is remains elusive, significant evidence exists for the involvement of an important enzyme designated terminal deoxynucleotidyl transferase (TdT). This unique DNA polymerase is overexpressed in ~90% of ALL patients. In addition, higher TdT levels correlate with poor prognoses, especially with respect to drug resistance. TdT is an exceptional therapeutic target as it is the only DNA polymerase that replicates single-stranded DNA rather than duplex DNA. This unique activity contrasts all other cellular DNA polymerases, and this distinction makes TdT an attractive target to develop selective nucleoside analogs to treat ALL.
Results of Biochemical Studies
Our group developed a novel nucleotide analog designated as 3-ethynyl-5-nitroindolyl-2′-deoxyriboside triphosphate. This novel compound functions as a potent chain-terminator against TdT activity. In addition, the unique chemical composition of this non-natural nucleotide prevents its utilization by other cellular DNA polymerases. These combined activities engender this nucleotide analog with exquisite selectivity for inhibiting TdT activity. The benefit of this selective inhibition is a lower risk of side effects typically observed with other anti-cancer nucleosides such as fludarabine, gemcitabine, and cordecypin that function as chain terminators for all DNA polymerases.
Results of Pharmacological Studies
The anti-cancer effects of the corresponding non-natural nucleoside (designated by its trade name, 5-Endosine) were tested against a panel of TdT-positive and TdT-negative leukemia cell lines. Our pre-clinical data shows an excellent correlation (>0.7) between the IC50 value of 5-Endosine with cellular levels of TdT in ALL cancer cells. In addition, the unique chemical composition of 5-Endosine allows for the covalent attachment of fluorogenic probes to the non-natural nucleoside inside cells. This technique provides a sensitive analytical method that can directly quantify the concentration of 5-Endosine in cancer and normal cells. This capability provides a unique diagnostic tool to gauge the therapeutic effectiveness of the nucleoside.
Preliminary toxicology studies performed in mice have evaluated the safety of 5-Endosine. These data confirm that mice treated with therapeutic concentrations of 5-Endosine do not show symptoms of toxic side effects such as diarrhea, anemia, leukopenia, and thrombocytopenia. While these results are encouraging, more detailed studies are being performed to confirm the safety of our compound.
Conclusion
5-Endosine analog possesses several pharmacological advantages over conventional nucleoside analogs. First, the non-natural nucleobase provides the perfect pharmacophore to selectively inhibit TdT activity. Secondly, since the deoxyribose moiety is left unperturbed, our analog possesses improved pharmacokinetic features such as efficient transport and metabolism. Finally, the ethynyl moiety allows for the efficient and selective attachment of fluorogenic probes to the non-natural nucleoside. The use of “click” chemistry provides a sensitive analytical method to quantify plasma concentrations of the nucleoside as well as the concentration in the nucleotide incorporated into the DNA of cancer cells.
Current Efforts
Our five-year goal is to perform Phase I and Phase II clinical trials on 5-Endosine. The first step in this process is to first file an Investigational New Drug (IND) application with the Food and Drug Administration (FDA). We have submitted several proposals to the National Cancer Institute (NCI) and other federal agencies to obtain funding to complete this work. Funds will be used for the synthesis of GMP-grade material that will then be used to evaluate the safety and toxicity profiles of 5-Endosine in two (2) animal models. We anticipate that these studies will be performed with the next 12 months.
